In welcome news for the research community, the Substance Abuse and Mental Health Services Administration (SAMHSA) is planning on proposing a rule to restore research access to substance use data in Medicare and Medicaid files.
The rule is nearing internal clearance, Pam Hyde, administrator for the [SAMHSA], said Friday. “We are having to work with the law and the regs and the lawyers about what it takes to get that and still be in line with the way the law is currently written,” Hyde said. A 1987 law requires express patient consent for health professionals to share substance abuse information. The law is unlikely to change soon—“most of us have determined that probably now is not the time to get Congress to change that,” Hyde said—so SAMSHA is chipping away at parts of it through regulation. Research is one use of the information that could be enabled through regulation.
This is terrific news. Austin and I have been encouraging SAMSHA to issue a proposed rule along these lines. The agency should be commended for pressing forward.
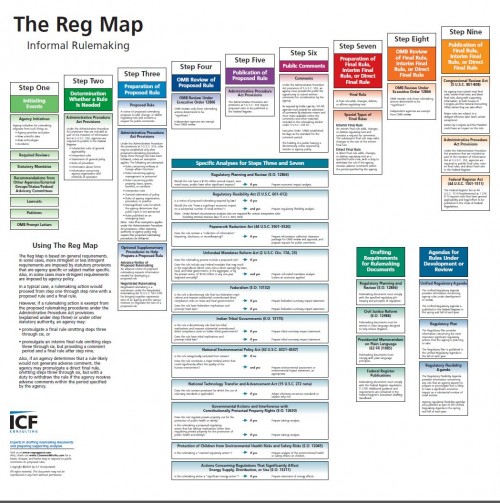
But it’s much too soon to declare victory. Shepherding a rule through the rulemaking process is almost comically difficult. Below is a stylized map of the typical rulemaking process (in reality, it’s even more complicated than this). Of the nine major steps, SAMHSA is on step three.
The rule could get derailed at any one of the next six steps. In particular, the rule could easily get stuck at step four: review at the Office of Information and Regulatory Affairs (OIRA), part of the White House’s Office of Management and Budget. In my prior academic work, I’ve written about how OIRA oversight of agency rulemaking puts a drag on rulemaking since any given rule can get entangled in bureaucratic or political snarls. That’s especially so when the rule raises sensitive questions about personal privacy.
So the research community needs to keep beating the drum—and keep beating it hard. To that end, Austin and I are on the hunt for more concrete examples of transformative research that would have been difficult or impossible without the data that CMS is now suppressing. To move the proposed rule from the drawing board to the Code of Federal Regulations, SAMHSA will need all the help it can get.