I recommend the Institute for Clinical and Economic Review’s technology assessment of proton beam therapy (PBT), which concludes,
We rated the net health benefit of PBT relative to alternative treatments to be “Superior” (moderate-large net health benefit) in ocular tumors and “Incremental” (small net health benefit) in adult brain/spinal and pediatric cancers. We judged the net health benefit to be “Comparable” (equivalent net health benefit) in several other cancers, including liver, lung, and prostate cancer, as well as hemangiomas. It should be noted, however, that we made judgments of comparability based on a limited evidence base that provides relatively low certainty that PBT is roughly equivalent to alternative therapies.
Also of note, PBT can cost at least twice as much the next best—or in many cases, comparable, and in some cases better—treatment modality, based on photons.
These are all points I’ve made before with respect to prostate cancer treatment. What’s new to me is the explanation of proton beam therapy physics in the report.
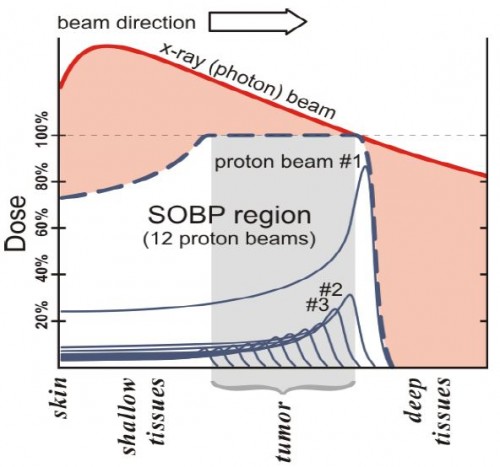
[P]rotons deposit radiation energy at or around the target, at the end of the range of beam penetration, a phenomenon known as the Bragg peak (Larsson, 1958). In contrast, photons deliver radiation across tissue depths on the way toward the target tumor and beyond, as depicted in [the figure] below. The total radiation dose for proton therapy is delivered in the “spread out Bragg peak” (SOBP) region from multiple proton beams; proton radiation is delivered to the target tumor as well as to shallow tissue depths before the target, but not to deeper tissue depths beyond the target (Levin, 2005).
This certainly suggests a strong, theoretical case for the superiority of protons. Of course, whether it plays out that way is an empircal issue. But, there are some theoretical concerns as well.
Some concerns have been raised about the hypothetical advantages of the radiation deposition for proton beams. The dose range is relatively certain for tumors that are close to the skin, but there is more uncertainty around the end of the dose range when deep-seated tumors such as prostate cancer are considered (Goitein, 2008). In addition, a penumbra (i.e., lateral spread or blurring of the beam as it reaches the target) develops at the end of the beam line, which can result in more scatter of the beam to adjacent normal tissue than originally estimated, particularly at deeper tissue depths (Rana, 2013). Protons are also very sensitive to tissue heterogeneity, and the precision of the beam may be disturbed as it passes through different types of tissue (Unkelbach, 2007).
Another concern is the effects of neutrons, which are produced by passively-scattered proton beams and result in additional radiation dose to the patient. The location of neutron production in a PBT patient and its biologic significance is currently a topic of significant debate (Hashimoto, 2012; Jarlskog, 2008). In addition, while it is assumed that the biologic effects of protons are equivalent to photons, specific relative biological effectiveness (RBE) values of protons in relation to photons are not known with absolute certainty for all types of tissues and fractionation schemes (Paganetti, 2002).
Since the report is ungated, you can click through to follow up on any citations of interest.