Not to be outdone by the Prime Minister, President Obama’s Council on Science and Technology met this morning to approve their report on preserving antimicrobial effectiveness (PCAST webcast here; report won’t be public for a few weeks; my unofficial transcript is here: PCAST Transcript; I live tweeted #PCAST).
Main points: we need surveillance, stewardship and new drugs. This is a “rate question” – we need to slow evolution and speed new drugs, balancing the two to protect public health.
Recommendations to the President in bold, with my comments in italics:
1. Stronger federal coordination on antimicrobial resistance, with cabinet-secretary level engagement and new responsibilities assigned to an existing White House office, overseen by a new Strategic Advisory Council. This seems a sensible plan, better than a new “bug czar” with no clear power.
2. Surveillance: increased funding for state and local surveillance, praising existing CDC efforts. New efforts will expand use of whole genomic sequencing as a surveillance tool. We are operating blind without comprehensive surveillance. Excellent idea.
3. New drugs:
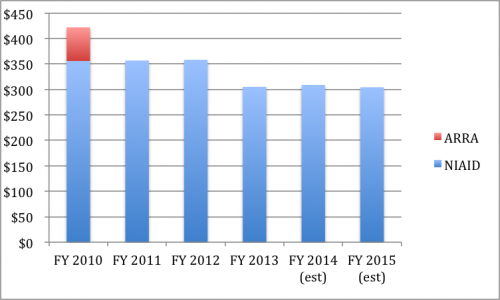
- Additional basic research funding. Absolutely essential. NIH funding for antibacterial resistance research is low and falling; we need to double real funding in this area over the next decade, giving researchers a sustainable research funding base.
US NIH Research Spending on Antimicrobial Resistance Research (FY 2010 – 2015, adjusted annually for US CPI, FY2010 base) (my data, not PCAST)
- Streamlined clinical trials. The FDA and EMA have already completed most of this work; this is an endorsement of that process. Antibiotic drug trial sizes are already quite small (n<200 in the treatment arm) and making them smaller will miss safety signals.
- Build a national infrastructure for abx trials. Perhaps like the Duke Clinical Trials initiative?
- “[T]remendous creativity” to develop alternatives to agricultural antibiotics. One way out of the political problem of agricultural use is to give US farmers cost-effective alternatives.
- New economic models to decrease cost and risk, including:
- BARDA-style grants. BARDA has been an important source of capital for antibiotic development and deserves more funding and a broader mandate.
- Advance Market Commitments. A model from vaccine procurement, not really R&D.
- Delinkage. Don’t pay for antibiotics based on volume because of resistance. This is the focus of most of my academic work lately.
- Tradeable vouchers. A easily grasped but wrong-headed idea.
- Prizes for diagnostics that shift clinical practice (mentions the UK Longitude Prize). A terrific idea, also one of the items modeled in the ERG Report.
- Educate policy makers and the public on the need for increased public investment in these “partial public goods.” Agreed. Continued antibiotic effectiveness are common pool goods, like fisheries.
4. Stewardship: It is “time to use the levers of the federal government [CMS] ” to support stewardship. Need to require stewardship programs in hospitals, reporting measures, make federal grants conditional on stewardship. Did not mention increased funding or a “billing code” for stewardship or infection control. Hospital CFOs see infection control as a cost center; help them do the right thing.
5. Agriculture: This is a politically sensitive topic. Report concludes that we need more data on just how much of the problem is from agricultural use, but says we still know enough to act now with “judicious use” on the farm. Praises recent FDA voluntary action on label revisions for growth promotion, with the potential for additional action later, after seeing results. I was hoping for at least a discussion of something stronger, like a user fee on agricultural use to fund further R&D, or the Preserving Antibiotics for Medical Treatment Act.
6. International cooperation: notes with approval the WHO Global Action Plan; did not mention the G7 process. The US has a global health security agenda for antibiotics. Right now, we have political mobilization at the highest levels ever seen. The US needs to build on that momentum and take Prime Mister Cameron up on his offer to address this at the G7 level, aggressively support this at the WHO; address resistance in the UN’s post-2015 development goals; and make it an important part of the global health security agenda. I should note that I’m a visiting fellow at the Chatham House Centre for Global Health Security, which has taken a leading role to address new business models for antibiotics, including the need for global cooperation.
@koutterson