COVID-19 is the novel coronavirus disease caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). This post will show that if we do not develop effective COVID-19 treatments, then there is a meaningful chance that this disease could kill many thousands of Americans. There is currently no such treatment, but one may be developed. This would be wonderful. However, antiviral drugs are expensive, and so is the hospital care required for severe COVID-19 cases. Treating this disease will stress American hospitals, and paying for costly treatments will likewise stress private insurance companies, Medicare, patients, and their families. These health system impacts will raise many fiscal, ethical, and legal questions. Epidemics move fast, and these questions will be challenging, so we should start discussing them now.
Suppose that a pharmaceutical company – perhaps Gilead Sciences, the maker of the antivirals Harvoni and Sovaldi – can quickly develop effective antivirals against SARS-COV-2. There are already several compounds that have been identified with activity, and rapid in vitro assays are available to test and refine compounds. Because COVID-19 is a life-threatening disease and demonstrating safety and efficacy won’t require large or long trials so the approval process would likely be accelerated. What the health policy community needs to begin discussing now is how these drugs should be deployed and managed. Initial supplies will likely be limited. How should these drugs be priced, who should get them, and how they should be paid for?
To project the cost of the epidemic, we first need to project how many people will get sick. This will depend on many factors, but we will consider just two. They are, first, what proportion of the population will get the disease (the Attack Rate) and how many of those who get the disease will die (the Case Fatality Rate, or CFR). Our understanding of these factors is uncertain, so we will present four scenarios involving different levels of these parameters.
We’ll consider two Attack Rates. In the first, 5% of the population gets COVID-19. This is the lower range of how many people get sick in seasonal flu. In the second, 19% of the population gets COVID-19. This is the rate of infection on the Diamond Princess cruise ship (700 of 3700 passengers).
The key facts about the COVID-19 CFR are that most people recover from the illness, but the rates increase with the patient’s age. The figure presents mortality data from a JAMA report on the Chinese experience in Wuhan.
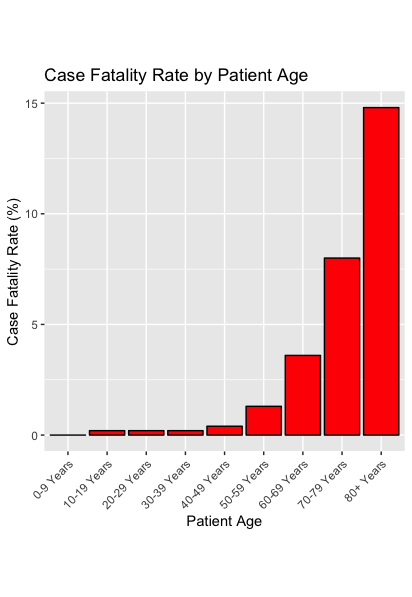
To project how many Americans might die of COVID-19, we consider two sets of CFRs. The first is the Wuhan CFR, presented above. However, it’s possible that Chinese health workers missed a significant number of COVID-19 cases. If so, the JAMA data would overstate the true CFR. Or it’s possible that the CFR will be lower in the US for some other reason. Therefore, we’ll also consider scenarios in which the age-specific CFRs are half what was reported for Wuhan. Using the 5% and 19% Attack Rates, and the Wuhan and Half-Wuhan CFRs, we’ll present four scenarios for the COVID-19 epidemic.
To project the number of deaths from COVID-19, we take the number of Americans in each age group, e.g., there are 11.9 million Americans aged 80 years and older. To get the number who develop COVID-19 if the Attack Rate is flu-like, we multiply that number by 5%, to get 595,548 COVID-19 cases. The full Wuhan CFR for this age group is 14.8%, so in this scenario, there will be 88,141 deaths in that age group. We sum across ages to get the total deaths for each scenario, presented below.
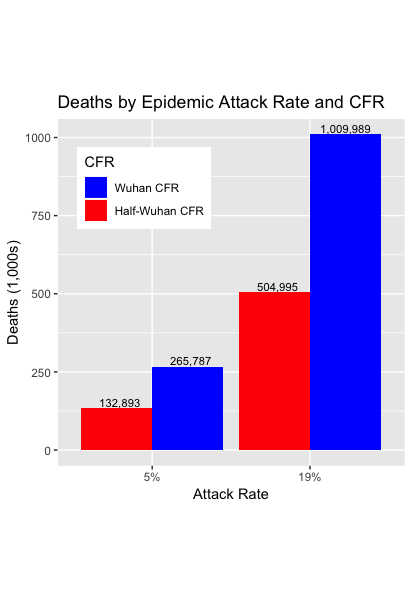
These numbers are shocking. Even under our most favorable scenario, roughly 133,000 people will die. Of greater concern, if we assume that US COVID-19 is half as lethal as the Wuhan data, then under a plausible 19% Attack Rate, there would be half a million deaths. Under the least favorable scenario, 1,000,000 will die.
These projections are extremely uncertain. The CFR for the US epidemic could be smaller than the Half-Wuhan scenario. But this uncertainty cuts both ways. It is also possible that more than 19% of Americans will be infected. More important, these are the projected deaths assuming that no new treatments are found.
We next consider what happens if an antiviral drug is developed that can save many of these lives. Most COVID-19 cases will not need to be treated. From the JAMA report on Wuhan:
Most [COVID-19] cases were classified as mild (81%; ie, nonpneumonia and mild pneumonia). However, 14% were severe (ie, dyspnea, respiratory frequency ≥30/min, blood oxygen saturation ≤93%, partial pressure of arterial oxygen to fraction of inspired oxygen ratio 50% within 24 to 48 hours), and 5% were critical (ie, respiratory failure, septic shock, and/or multiple organ dysfunction or failure).
We’ll assume that the proportion of cases that are severe enough to be treated will be twice the CFR for each age group. So, for the 80 and older group in the full Wuhan CFR scenarios, we would expect to treat 2 × 14.8% = 29.6% of the infected persons.
Now, suppose that pharmaceutical researchers develop antiviral drugs that are effective in treating COVID-19. The treated cases will be hospitalized and get a course of antivirals. How much will a course of antiviral treatment cost? From a pharmaceutical company’s perspective, this drug would be bringing substantial value to patients and society, not all that different from Sovaldi, the infamous $1000/pill drug, and it wouldn’t be surprising if they sought to establish a similar price point. Let’s suppose that the price of a course of antivirals is $20,000. How much will the hospital care cost? From various sources, we speculate that each severe case will need 17 days in the hospital, 5 of which will be in an ICU. Assuming $10,000/day for an ICU day and $1,000/day for a regular hospital day, that is $62,000 for each hospitalized case. Taking all this into account, the next Figure shows the treatment costs (that is, the combined antiviral and hospital costs) for the four scenarios.
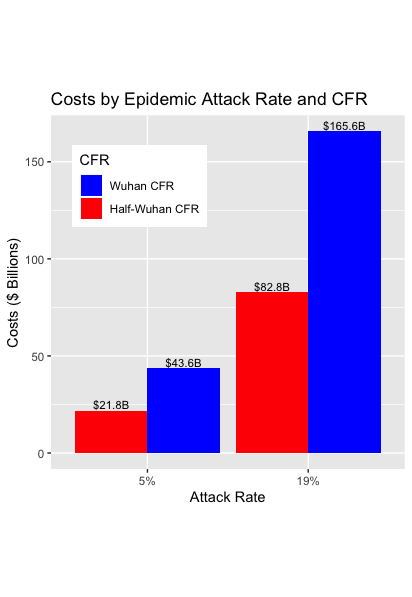
The figures here are, again, shocking. The projected costs of anti-viral treatments and hospitalizations range from $22 to $166 billion.
Again, these are rough and ready projections. They exclude the extensive additional costs for testing for SARS-CoV-2 and for efforts to prevent the disease. But these staggering costs raise several questions.
1. Who will pay for COVID-19 treatment? These costs could bankrupt private health insurance plans. However, the majority of patients are likely to be covered by Medicare. In FY2018, the Medicare budget was $593 billion. So, the costs of COVID-19 treatments under our scenarios run from 4% to 28% of the Medicare budget. As we write, there are accountants at the Department of Health and Human Services who are having trouble sleeping. Finally, there will be some persons younger than 65 who need treatment, among whom there may be thousands of uninsured and underinsured persons facing what we project to be more than $80,000 of treatment costs.
2. Where will we find the hospital and ICU beds? There is concern that the US lacks adequate hospital surge capacity to handle a pandemic. (Canada, which runs its hospitals close to full capacity, may be at even greater risk.) The Wuhan experience indicates that if an ICU bed is not available, patients with severe COVID-19 will die. The health care system in Wuhan was overwhelmed, and that’s a reason why the CFR in Wuhan was substantially higher than in other provinces or outside of China. We have about 70,000 ICU beds in the US, but these are usually in use. If there are many cases and they cluster regionally, the system will be overwhelmed in some locations. If that happens, the CFRs will increase. We can hope that an effective antiviral will prevent most of these severe cases, but this only helps if antivirals are in good supply and people can afford to take them.
3. If ICU beds and courses of antivirals are in limited supply, how do we ration them? On what principles would treatments be allocated? Who is empowered to make rationing decisions? If the President and/or the Congress have that power, how confident are we that they will be able to make what could be the most controversial health care policy decision in American history?
4. Should we pay what a pharmaceutical company will want to charge for antivirals? Our projections indicate that the companies would receive many billions of dollars of revenue. They also suggest that the costs of treatment will place great fiscal stress on public and private insurers, to say nothing of individuals who lack adequate coverage. If we don’t want to pay what the industry wants to charge, what’s the legislative or legal mechanism to get a better price?
5. Will these hypothetical COVID-19 treatments be cost-effective? And should we even think about that question?
David J. States, Bill Gardner, and Nicholas Bagley
David States, MD, PhD, is an internist and a computational biologist. He is the Chief Scientific Officer for Angstrom Bio.
