Generic entry for small molecule drugs is controlled by Hatch-Waxman, allowing generic entry 10 – 20 years after FDA approval. For monoclonal antibodies and other large molecule biological products, a different law applies, which may never result in effective generic competition, even after patent expiration.
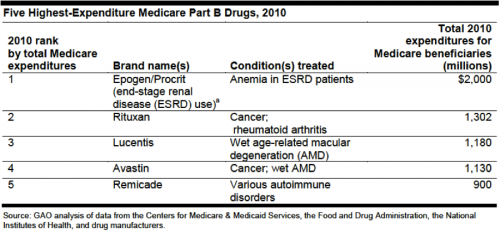
Can you guess the top 5 drugs are in Medicare Part B? None are small molecule drugs subject to Hatch-Waxman in the US:
Effective generic competition may never trouble these drugs. Indeed, Amgen’s base patent on epoetin alfa expired years ago. Payment reform should be considered, as a backstop to the biosimilar pathway.
n.b. – this data is Part B, not C or D.
@koutterson