The following is coauthored by Vivian Ho, Ph.D. (@healthecontx), James A. Baker III Institute Chair in Health Economics, Kirstin Matthews, Ph.D. (@stpolicy), Baker Institute Fellow in Science and Technology Policy and Heidi Russell, M.D., Ph.D., Associate Professor, Department of Pediatrics, Baylor College of Medicine and Associate Director, Center for Medical Ethics and Health Policy, Baylor College of Medicine.
The May 24 release of This Week in Virology cited Winston Churchill’s famous words after winning The Battle of Egypt: “Now this is not the end. It is not even the beginning of the end. But it is, perhaps, the end of the beginning.” We couldn’t agree more. This week we compare actions to address the pandemic across countries, the damage inflicted by the coronavirus across states, and we provide a detailed summary of vaccine development.
By the Numbers
As of Friday, May 29, data from the Covid Tracking Project showed that the 7-day average (smoothed) number of new U.S. daily cases fell to 20,384, a 10.7% drop from 22,570 a week earlier. The percent of cases testing positive fell from 6.2% a week earlier to 5.3% last Friday, although the 382,922 tests for the nation still lie well below the target of 900,000 tests per day. Here in Texas, the smoothed number of cases was relatively constant at 928 last Friday, compared to 921 a week ago. The smoothed number of cases in Texas had mostly been falling since mid-May, but reports of multiple packed night clubs over Memorial Day weekend likely halted the state’s successful battle against the coronavirus. Testing remained flat over the week, with 22,317 tests on Feb 15 and 22,525 last Friday.
Limited U.S. Capacity for Testing
In the seventh Covid-19 webinar hosted by the American Public Health Association and the National Academy of Medicine, former FDA Commissioner Scott Gottlieb stated that the U.S. could only expect to be able to conduct 500,000 viral tests per day using the current approach, which requires specialized nasopharyngeal swabs and reagents. Manufacturing supply chain constraints will prevent us from surpassing that level. We found it remarkable, then, that the city of Wuhan recently tested 9 million of its 11 million people in just 10 days. On Friday May 22, they tested 1.47 million people. The city employed “sample pooling,” where individual samples from five to 10 people were tested together in a single tube. This approach suggests that the city processed 1 million+ tests in 10 days. If U.S. testing capacity is only slightly larger than one city in China, we don’t have much hope for avoiding a second wave of infections in the coming months.
Vaccine Development
On May 28, the Baker Institute Center for Health and Bioscience hosted a discussion on the Covid-19 vaccine with Dr. Peter Hotez and Rekha Lakshmanan. In addition, many of these issues and the U.S. plan were reviewed in an essay for Science by Dr. Anthony Fauci and NIH director Dr. Francis Collins. Below are some highlights from the discussion.
Vaccines Development and Approval
- The vaccines currently under clinical trials from Oxford University, Moderna, and Inovia all employ new techniques, which allow them to be developed or tested faster than traditional vaccines, which use either killed or attenuated (weakened) virus.
- It is uncertain if these novel vaccines will create enough of an immune response to be effective. Dr. Hotez worries that these vaccines will not create enough neutralizing antibodies, which prevent the virus from entering cells and therefore replicating.
- The September date being floated in the press is just a potential date when manufacturing of potential vaccines could start, not a realistic date that a vaccine will be approved for use. Data from the clinical trial that would tell us if the vaccine is safe and effective could take six months to a year.
- The current vaccine strategy pushes several different candidates simultaneously, perhaps more than a dozen, to see which works best. Manufacturing can begin for vaccines that reach Phase 3 trials, so that an approved vaccine can be quickly disseminated. However, 90% of vaccines fail, so there is a risk of spending funds on vaccines that will never be put to public use.
- The first vaccines might not be highly effective at preventing the disease, but could serve as preliminary vaccines until a more effective vaccine is available.
Controlled Infection Trials
- Policymakers have discussed controlled infection trials for Covid-19 (previously discussed here), which would purposely expose volunteers to the SARS-CoV2 virus to see if the vaccines work.
- However, protocol development is complicated. One needs large scale, safely manufactured viruses, and one must determine the ideal quantity of virus to challenge the vaccine, but not cause serious illnesses. The lack of treatment for Covid-19 also increases the risk of exposing individuals to the virus.
Community Immunity
- To move past this pandemic we need to have herd immunity or “community immunity.” Estimates for community immunity for Covid-19 are at least 70% — which means at least 70% of the population (or 200 million Americans) would have to be exposed to the virus.
- We are unlikely to achieve community immunity without a vaccine.
- Lakshmanan noted that we will have to find a balance between being pragmatic and cautious, but she encouraged that leaders be transparent with their plans with clear expectations.
The State-by-State Death Toll and Economic Toll
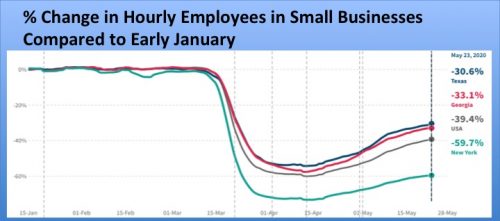
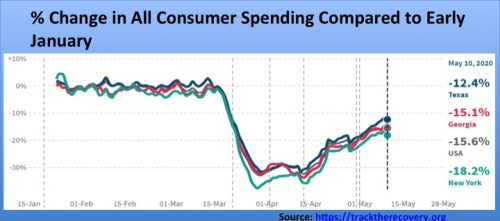
Opportunity Insights, a research team led by Harvard economist Raj Chetty, has published an Economic Tracker, which allows one to readily compare the economic toll of Covid-19 across states. The data is not as comprehensive as data from the U.S. Census Bureau, but it is more recent. The graphs below depict the devastation the pandemic has played on small business employment and consumer spending in the U.S. and three states. In New York, with 151 deaths per 100,000 as of May 26, small business employment was down 59.7% compared to early January. Texas and Georgia had five and 18 deaths per capita, respectively. These two states relaxed lockdown restrictions relatively quickly, yet small business employment is still down 30.6% and 33.1%, respectively. The magnitude of falls in consumer spending are smaller, but still in the double digits. States need to work harder at further reducing Covid-19 prevalence in their communities, before many consumers will feel safe to resume regular spending at small businesses such as restaurants, retail shops, hair salons and gyms.
There Must be a Better Way
Fear of coronavirus led many New York residents to avoid their subway system in mid-March, and interest in car buying to avoid public transportation has surged around the world. We contrast these actions with a photo in last week’s Wall Street Journal of masses of masked passengers preparing to board a Hong Kong subway train, with no apparent social distancing. As of Friday, the U.S. reported 18,913 new COVID-19 cases. Singapore reported 373, Japan 32, and Hong Kong 1. The WSJ article summarized Hong Kong’s and Singapore’s playbook: test widely, quarantine aggressively and treat patients early. Keeping infections from spreading to communities most at risk allowed both cities higher hospital capacity to treat patients. One expert said, “It’s not too late. [U.S.] states can still learn from this model.”
Other Developments
Random antibody testing of 4,600 people in Indiana suggests that 3% (188,000) of the state’s population have been infected with the coronavirus, supporting previous studies suggesting that the mortality rate for COVID-19 is roughly 0.5%.
Two new studies show that current and former cancer patients who developed COVID-19 were much more likely to die within a month than people without cancer who got it.
At least 35% of people who contract COVID-19 are asymptomatic. Although they appear to be less infectious to other people, a study in Wuhan indicates that asymptomatic people could still shed the virus for 8 days.
The American Hospital Association estimates a total four-month loss of $202.6 billion for hospitals and health care systems because of excess costs for COVID patients and cancelled non-emergent care. Becker’s Hospital Review is maintaining a running list of announcements of hospital closures, furloughs, and layoffs. In an earlier blog we predicted that financial losses will differ by hospital types and location. Fair Health found that larger hospitals (over 250 beds), especially in the northeastern US, suffered the largest revenue drops in the first quarter of 2020.
Outpatient healthcare decreased nearly 60% in March. According to the Commonwealth Fund and Phreesia, a company that manages clinic scheduling software, in-person outpatient visits are increasing and are now about 2/3 of pre-pandemic levels. Decreased regulations on telehealth allowed providers and patients new opportunities to interact. Telehealth visits seem to have peaked and even declined somewhat, but telehealth will likely be a permanent fixture in outpatient care.
The End of the Beginning
As most states have begun reopening their economies, we have reached the end of the beginning, with many more months to come in the COVID-19 pandemic. We began this weekly blog as a means to learn about the COVID-19 virus, how it spreads, and what scientists and policy makers could do to end the pandemic. We conclude from our analysis that those states and countries that invest aggressively in testing and contact tracing will have the best chance at beating the virus. Time will tell, but early data leads us to hypothesize that effective testing and contact tracing will also lead to the strongest and fastest economic recovery. We will continue to update you on treatment and vaccine developments, but we are transitioning to an abbreviated weekly update, as we must return to our other research obligations. We look forward to celebrating with you in person when we reach the beginning of the end.